Overview - Why is it important to check your cell line(s) for Mycoplasma contamination?
Mycoplasma are bacteria that can grow in cell cultures without any visual indication of their presence. It has been estimated that 10 to 30% of cell lines are infected with Mycoplasma leading to severe biochemical and laboratory consequences.
What is Mycoplasma?
Mycoplasma are very small bacteria of approximately 0.1 µm in diameter. They are highly flexible bacteria without cell walls. More than 100 Mycoplasma species are known and common Mycoplasma contaminants are Mycoplasma arginini, M. fermentans, M. orale, M. hyorhinis, M. hominis, M. genitalium, M. salivarium, M. synoviae, M. pirum, M. gallisepticum, M. pneumoniae, M. yeatsii, Spiroplasma citri and Acholeplasma laidlawii.
For more details about Mycoplasma, see the article Mycoplasma Contamination.
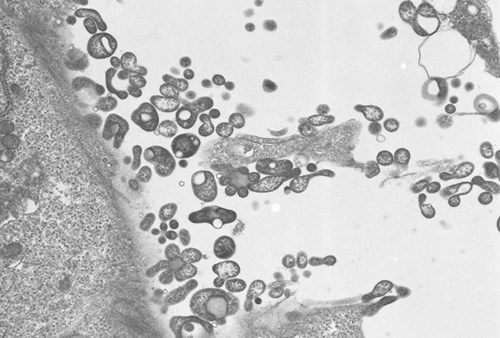
Why is Mycoplasma a problem in cell cultures?
The detection of Mycoplasma poses a challenge for researchers, as a Mycoplasma contamination is not visible under the light microscope. Mycoplasma can achieve very high densities with 107 to 108 organisms per mL and without changes to medium pH or medium turbidity.
Additionally, Mycoplasma contamination cannot be removed by filtration and the organisms are resistant to most antibiotics.
What are the consequences of Mycoplasma contamination?
Mycoplasma contamination of a cell culture can have severe biochemical consequences
such as:
- Inhibition of cell proliferation
- Alteration of cell behaviour and response
- Inhibition of RNA and protein biosynthesis
- Contamination with mycoplasma proteins and DNA in extractions
- Alteration of immunological reactions
- Alteration of microarray gene expression profiles
- Alteration of virus proliferation and infection rates
- Resulting in erroneous data and misleading publications
- Subsequently a potential loss of reputation
Mycoplasma contamination of a cell culture can also have severe laboratory consequences such as:
- Loss of cells and entire cell cultures and products
- Loss of research time and money
- Additionally, Mycoplasma contaminations are a biosafety concern
Applications – Guidlines of journals such as BioMed Central (BMC)
When human cell lines are used for investigations that are going for publication, authors are strongly encouraged to include the following information in their manuscript:
- The source of the cell line, including when and from where it was obtained
- Whether the cell line has recently been authenticated and what method was utilised for authentication
- Whether the cell line has recently been tested for Mycoplasma contamination
Products related to Mycoplasma contamination testing
With Mycoplasmacheck, researchers can conveniently let their cell cultures be tested for Mycoplasma contamination. Mycoplasmacheck is service based on qPCR for quick and precise test with standardised protocols and automated workflows in an accredited laboratory.
Mycoplasmacheck uses specifically designed primers and probes for the detection of all Mycoplasma species that commonly contaminate tissue culture.
Workflow - Methods & technology for Mycoplasma contamination test
Sample for Mycoplasmacheck can be taken without additional effort at every cell culture media change:
- Sample preparation takes less than 15 min.
- Samples are conveniently shipped through any Eurofins Genomics DropBox or shipped separately.
- The standardised qPCR test for Mycoplasma is performed according to the ISO17025 accreditation.
- All Mycoplasma species in the European Pharmacopoeia (E.P) 2.6.7 are detected plus approximately 100 additional mollicutes (bacteria without cell wall).
- The test has no cross-reactivity with E.coli, B. subtilis, P. aeruginosa, P. mirabilis or S. aureus.
- The test does not detect eukaryotic DNA, e.g. from human, mouse, hamster or bovine origin.
Did you know that we at Eurofins Genomics have great expertise in Sanger sequencing that converges into a comprehensive product portfolio with services tailored to your needs such as the Mix2Seq service and TubeSeq service?